Phase 3 Readout Of Daraxonrasib In 2L Metastatic PDAC Marks An Inflection Point For Pancreatic Cancer
Median OS 13.2 vs 6.7 months, HR 0.40, in pretreated metastatic pancreatic ductal adenocarcinoma against standard chemotherapy. The era of targeted therapy in pancreatic cancer has finally arrived.

Disclosure: The author holds beneficial long positions in Revolution Medicines, Inc. (NASDAQ: RVMD) and Erasca, Inc. (NASDAQ: ERAS). This article is provided for informational and educational purposes only and is not financial advice. The illustrative survival model discussed here is a closed-form Weibull proportional-hazards mapping parameterised from two reported topline endpoints, not a fit to patient-level data. It cannot reliably predict the outcome of any real-world study, subgroup, or individual patient. Although the author is a physician, this content represents a personal analytical perspective on the companies' science and does not constitute medical advice, a diagnosis, or a treatment recommendation. The author receives no compensation for this article and has no business relationship with the companies mentioned. Please see the full "Legal Information and Disclosures" section below.
Pancreatic ductal adenocarcinoma (PDAC) still has a dismal five-year relative survival of 13% (Siegel et al., 2026), while survival in other solid malignancies has improved dramatically. Breast cancer now reaches a five-year survival of 91%, prostate 98%, colorectal 65%, lung 28% (Siegel et al.), each reshaped over decades by targeted therapies: HER2-directed agents in breast cancer, anti-VEGF and anti-EGFR agents in colorectal cancer, EGFR and ALK inhibitors in lung cancer. PDAC never got its counterpart. Systemic therapy still relies on cytotoxic chemotherapy.
Each successive regimen since gemcitabine monotherapy in 1996 (FOLFIRINOX, gemcitabine plus nab-paclitaxel, NALIRIFOX) has been a more toxic, untargeted combination rather than a new mechanism. Even after curative-intent resection and adjuvant mFOLFIRINOX, 5-year DFS was only 26.1% in PRODIGE 24 (Conroy et al., 2022): roughly three in four patients relapse, because adjuvant chemotherapy does not reliably eliminate the micrometastatic deposits that seed recurrence.
More than 90% of PDAC tumors carry an oncogenic RAS mutation (Halbrook et al., Cell 2023), dominated by KRAS G12D, G12V, and G12R. RAS was the paradigmatic undruggable oncogene: picomolar GTP affinity foreclosed competitive inhibition, and its surface was considered too smooth to engage. Sotorasib cracked KRAS G12C in NSCLC in 2021 (Skoulidis et al., 2021) through a reactive cysteine in the switch II pocket, accessible only when the protein is in the GDP-bound OFF state. G12C works as an OFF-state target because it retains an unusually fast intrinsic GTPase activity that keeps refilling the OFF-state pool the drug can trap. The dominant PDAC mutations, G12D and G12V, have slower GTP hydrolysis and sit predominantly in the active GTP-bound ON state, where OFF-state inhibitors have no purchase. And G12C drives only 2% of PDAC regardless.
Daraxonrasib (RMC-6236) is an oral non-covalent tri-complex inhibitor: it binds the intracellular chaperone cyclophilin A, and the resulting binary complex engages the ON state of RAS directly, sterically blocking its interaction with downstream effectors such as RAF (Holderfield et al., 2024; Jiang et al., 2024; Wasko et al., 2024). Because it targets the signalling-competent state shared by most RAS variants, a single molecule covers G12X, G13X, and Q61X mutations across KRAS, NRAS, and HRAS, reaching the driver in roughly nine in ten PDAC tumors.
RASolute 302 randomised patients 1:1 to daraxonrasib 300 mg orally once daily or investigator's choice of standard-of-care cytotoxic chemotherapy in previously treated metastatic PDAC, across the full RAS spectrum including wild-type tumors. The April 13, 2026 topline reported median OS of 13.2 vs 6.7 months, HR 0.40, p < 0.0001, with a concordant PFS benefit, crossing the pre-specified boundary at the first interim analysis. The historical benchmark is NAPOLI-1 (Wang-Gillam et al., 2016): HR 0.67 and median OS 4.2 to 6.1 months, unmatched in second-line since. HR 0.40 from an oral monotherapy against modern combination chemotherapy, in patients already progressed on FOLFIRINOX or gemcitabine plus nab-paclitaxel, is not on the same continuum as any prior advance in this disease.
The reaction to the Phase 3 readout has been overwhelmingly enthusiastic, but I have also seen posts on X dismissing it: another 6.5 months on the median, how much does that really help patients? For readers who don't routinely think in terms of median survival, this framing obscures more than it reveals. What HR 0.40 implies for individual patients is worth pausing on, because the reported median understates the experience of those who respond. Figure 1 is a closed-form illustration: a Weibull-PH model with common shape, forced to match both the median ratio (13.2/6.7) and HR (0.40), pins the shape parameter analytically at k ≈ 1.35 with no free parameters.
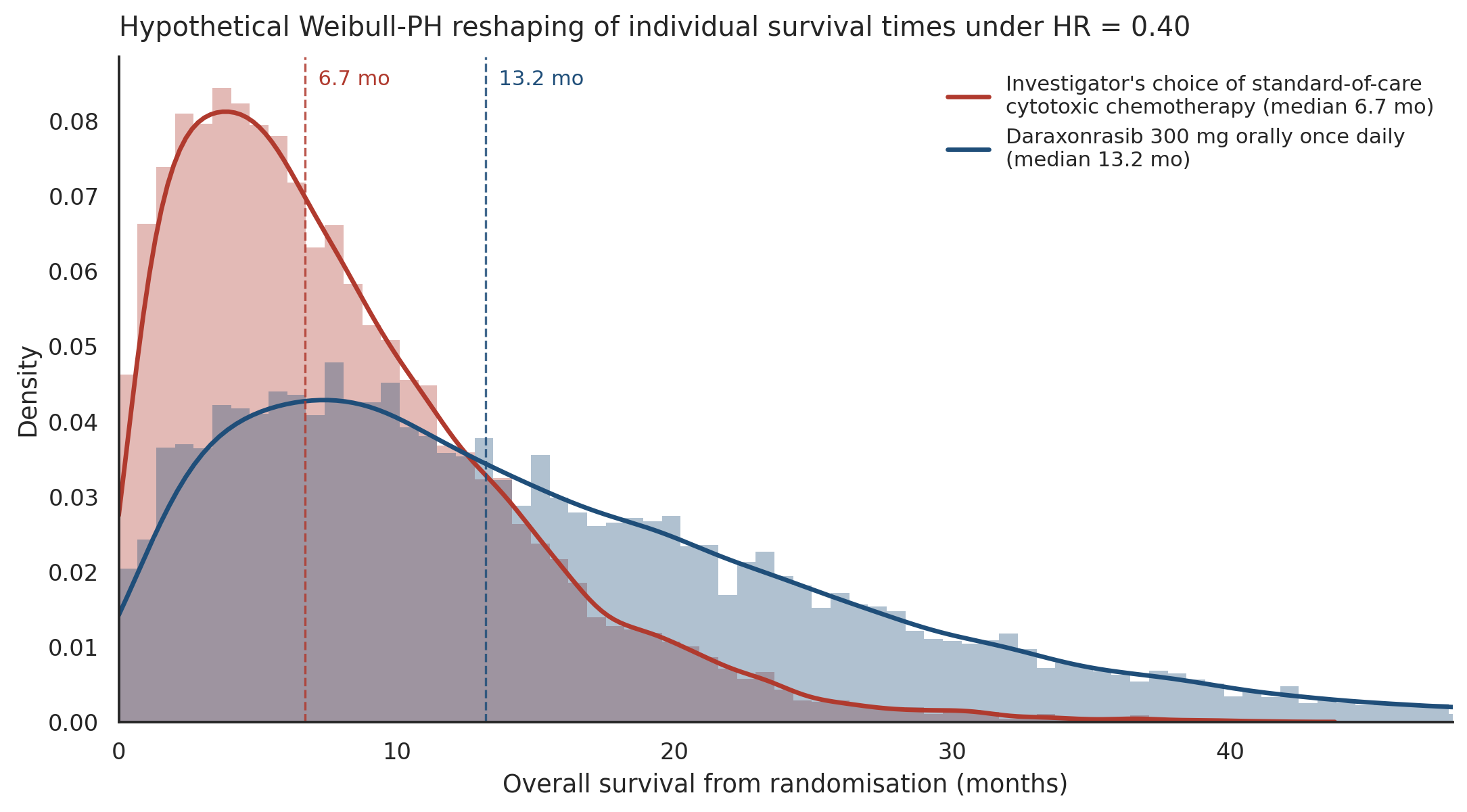
Under this model, the proportion of patients alive at 12 months rises from 22% on chemotherapy to 54% on daraxonrasib, from 7% to 35% at 18 months, and from 2% to 21% at 24 months. For pretreated metastatic pancreatic cancer, where being alive at two years has been a statistical rarity, a roughly tenfold increase in the probability of reaching that landmark is not an abstract improvement. Whether the Kaplan-Meier curves, likely presented at ASCO 2026, compress around the median or open into a durable-responder tail, both departures from this model, is a question for longer follow-up.
What makes this readout different from prior advances in PDAC is that three things happened at once. The effect size halves the best hazard ratio previously achieved in second-line PDAC, from 0.67 in NAPOLI-1 to 0.40 here. The mechanism is, for the first time in this disease, a targeted therapy reaching the driver in more than nine in ten tumors rather than a more intensive chemotherapy combination. And the modality is an oral once-daily pill rather than an infusional cytotoxic regimen.
RASolute 302 shows what daraxonrasib can do in second-line metastatic disease. Two Phase 3 trials test the same mechanism earlier. RASolute 304 (first patient dosing disclosed December 18, 2025) compares daraxonrasib against observation after surgical resection and neoadjuvant and/or adjuvant chemotherapy in resectable RAS-mutant PDAC, with disease-free survival as the primary endpoint. The adjuvant setting is biologically favourable: the micrometastatic deposits that seed recurrence are small, dispersed, not yet shielded by fibrotic stroma, and RAS-dependent given that the primary tumor was RAS-mutant.
How much an adjuvant benefit could move aggregate PDAC five-year survival comes down to stage and resectability arithmetic. In SEER 2015-2021 data, 14.6% of PDAC patients present with localised disease (43.6% 5-year survival), 29.8% regional (16.2%), and 49.3% distant (3.3%). Clinically, roughly 15 to 20% of new diagnoses are upfront-resectable, another 10 to 15% borderline-resectable (where resection is feasible only after neoadjuvant downstaging), around 30% locally-advanced, and about half metastatic at presentation. Improving outcomes in metastatic disease, even by a large relative margin, moves aggregate five-year survival only modestly, because the stage-specific base rate is so low that very few patients cross the five-year threshold from that subgroup. Reducing recurrence after curative-intent surgery operates on the highest stage-specific base rate, and could, if durable, move resected patients' outcomes toward the range adjuvant targeted therapy has produced in other diseases. But this lever reaches only those patients who make it to resection: the upfront-resectable group, and the subset of borderline-resectable and locally-advanced patients who respond sufficiently to neoadjuvant or induction therapy to become surgical candidates.
This is where the neoadjuvant setting becomes the complementary lever. A tolerable oral targeted therapy could, if radiographic responses prove deep enough, downstage a share of borderline-resectable and locally-advanced tumors to resectability, expanding the fraction of patients who reach curative-intent surgery and, downstream, who benefit from an adjuvant RAS inhibitor. Whether daraxonrasib's response pattern in metastatic disease will translate this way is not yet established and requires dedicated trials. The most important caveat to the broader argument is that the adjuvant trial's primary endpoint is disease-free survival, and a DFS benefit does not automatically translate into a five-year overall survival benefit: durability over years is what would actually move the aggregate numbers.
RASolute 303 (first patient dosing disclosed April 2, 2026) is the first-line metastatic Phase 3 trial, comparing daraxonrasib monotherapy, daraxonrasib plus gemcitabine and nab-paclitaxel (GnP), and GnP alone, irrespective of RAS status. Phase 1/1b first-line cohorts showed ORR 47% / DCR 89% on monotherapy (n=38) and 55% / 90% on the combination (n=31). Whether daraxonrasib alone can match or exceed a chemotherapy-containing combination in the first-line setting, and whether adding chemotherapy to daraxonrasib meaningfully improves on either single modality, are open questions the trial is designed to answer.
Daraxonrasib is the first but not the only tri-complex RAS(ON) inhibitor in clinical development. Revolution Medicines has allele-selective candidates further back in its pipeline: zoldonrasib (G12D-selective) received FDA Breakthrough Therapy Designation in G12D-mutated NSCLC in January 2026, and G12D is the most common KRAS variant in PDAC at roughly 40% of cases (Halbrook et al.); elironrasib (RMC-6291) is G12C-selective; and RMC-5127 (G12V-selective) had its first patient dosing disclosed on January 29, 2026. Revolution Medicines has disclosed plans to initiate RASolute 309 in 2H 2026, a Phase 3 trial of the daraxonrasib-plus-zoldonrasib doublet in first-line metastatic PDAC, reflecting a strategy of using daraxonrasib as a multi-selective backbone with allele-specific reinforcement. Erasca has entered the class in Phase 1 with ERAS-0015, another cyclophilin-A-based pan-RAS molecular glue, with initial monotherapy data expected in H1 2026.
With RASolute 302, pancreatic cancer joins the list of solid tumors in which a targeted therapy has produced a substantial survival benefit against standard-of-care chemotherapy in Phase 3. The next several years of readouts will show whether daraxonrasib and the allele-selective agents behind it can go further: earlier in the disease, in combination, and over longer follow-up, toward the kind of long-term survival changes that targeted therapies produced in other solid malignancies such as breast, colorectal, and lung cancer. RASolute 303, RASolute 304, and RASolute 309 are the immediate tests, with RASolute 304 plausibly carrying the largest potential to move aggregate five-year survival if the benefit proves durable: after resection, residual disease is likely micrometastatic, no longer shielded by the primary tumor's stroma, and potentially more amenable to durable suppression than the metastatic setting in which daraxonrasib has already succeeded. RAS is now druggable in pancreatic cancer. This is the inflection point.
Follow me on X for frequent updates (@chaotropy).
Legal Information and Disclosures
General Disclaimer & No Financial Advice: The content of this article is for informational and educational purposes only. It represents the personal opinions of the author as of the date of publication and may change without notice. The author is not a registered investment advisor or financial analyst. This content is not intended to be, and shall not be construed as, financial, legal, tax, or investment advice. It does not constitute a personal recommendation or an assessment of suitability for any specific investor. This article does not constitute an offer to sell, a solicitation of an offer to buy, or a recommendation of any security. Readers should conduct their own independent due diligence and consult with a certified financial professional before making any investment decisions. This article is not a securities research report and has not been prepared in accordance with legal requirements designed to promote the independence of investment research.
Medical Disclaimer: Although the author possesses a medical background, the information presented here regarding clinical trials, survival endpoints, or pharmaceutical mechanisms is strictly for the purpose of educational discussion and general commentary regarding the underlying science. It does not constitute medical advice, a diagnosis, or a treatment recommendation, nor does it establish a physician-patient relationship. Readers should never disregard professional medical advice or delay in seeking it because of something read on this website. Always consult a qualified healthcare provider regarding any medical condition.
Illustrative Model Disclaimer: The figure in this article is a hypothetical illustration, not a fit to patient-level data. It is a closed-form Weibull proportional-hazards mapping, parameterised by the two reported RASolute 302 topline endpoints (median OS on each arm and the hazard ratio), and is intended only to build intuition for what HR 0.40 implies for the shape of a survival distribution. The simulation code has not been independently audited, peer-reviewed, or verified by a third party. Bugs in the implementation could alter results materially. The model's outputs should be understood as a structurally conservative illustration, not as quantitative predictions of survival on either arm, in any subgroup, or in any individual patient. The model makes no claim about the existence or size of a durable-responder tail, which can only be assessed from the actual Kaplan-Meier curves with longer follow-up.
Accuracy and Third-Party Data: Clinical trial data, press releases, regulatory filings, and published literature referenced in this article are sourced from ClinicalTrials.gov, company press releases, peer-reviewed journals, and conference abstracts. While the author believes these sources to be reliable, the completeness, timeliness, or correctness of this data cannot be guaranteed. The author assumes no liability for errors, omissions, or the results obtained from the use of this information.
Disclosure of Interest: The author holds beneficial long positions in Revolution Medicines, Inc. (NASDAQ: RVMD) and Erasca, Inc. (NASDAQ: ERAS). The author reserves the right to buy or sell these securities at any time without further notice. The author receives no direct compensation for the production of this content and maintains no business relationship with either company. The author has no access to non-public trial data, unblinded interim results, or internal company information. Readers should be aware that the author's financial interests in these companies create a potential for confirmation bias in framing, selection of comparators, and interpretation of forward-looking implications, despite efforts to mitigate this through conservative parametric choices, hedged language, and transparent disclosure of the model's limitations. Confirmation bias can manifest in subtle ways, including the choice of historical benchmarks, the selection of which adjacent trials to emphasise, and the tone of discussion around unproven settings. Independent replication of the model with alternative assumptions is encouraged.
Forward-Looking Statements & Risk: This article contains forward-looking statements regarding regulatory outcomes (such as FDA decisions), clinical results, pipeline development, and potential population-level effects on cancer survival statistics. These statements are predictions based on current expectations and are subject to significant risks and uncertainties. Actual results may differ materially. Investing in biotechnology and pharmaceutical securities involves a high degree of risk, including the potential for total loss of principal. Past performance is not indicative of future results. No reader should rely on this article as a basis for any investment decision. The author has no obligation to update this article if new information becomes available or if the author's position changes. This content is not directed at or intended for distribution to any person or entity in any jurisdiction where such distribution would be contrary to law or regulation.
Copyright: All original content, including text and figures, is the property of the author and may not be copied, reproduced, or published, in whole or in part, without the author's prior written consent, except as permitted by applicable law or the terms of the platform on which it is published. Use of this content for training machine learning or AI models is not permitted without explicit authorization. Third-party or public domain images remain subject to their respective rights and are not claimed as the author's property.